Activity is an effective concentration, based on something callef chemical potential. The concentration of a solution is a measure of how much stuff is dissolved in a liquid. Activity means effective concentration of matter which ready to react. What is difference between activity and concentration?Ĭoncentration means the total amount of product or reactant. The activity series of the halogens is fluorine > chlorine > bromine > iodine. Since metals replace other metals, while nonmetals replace other nonmetals, they each have a separate activity series. The activity series is a list of elements in decreasing order of their reactivity. What is activity series of metals and non metals? In chemical thermodynamics, activity (symbol a) is a measure of the “effective concentration” of a species in a mixture, in the sense that the species’ chemical potential depends on the activity of a real solution in the same way that it would depend on concentration for an ideal solution. Which is the most reactive in the activity series of metal?Ĭaesium, the most reactive metal in the periodic table, reacts extremely violently – hence why it can’t be demonstrated in a classroom! This can be compared to other common metals, such as iron and copper, which produce no reaction when dropped into water. During displacement reactions, a more active nonmetal displaces a less active nonmetal from a compound. Similar to metals, we can also arrange nonmetals in terms of their reactivity. Nonmetal activity series is a list in which nonmetals are arranged in the decreasing order of their reactivity. What is the activity series of non metals? 
How can activity series be used to predict reactions? How is the experiment done to determine the activity series of metals? The mnemonic to remember the reactivity series of metal is : Please send cats, monkeys and zebras in lovely happy cages made of silver, gold & platinum.The reactivity series is the series of metals arranged in decreasing order of their reactivity..For nonmetals, greater activity means a greater ease of gain of electrons, to form negative ions. For metals, greater activity means a greater ease of loss of electrons, to form positive ions. What is meant by the activity of an element?Īn element’s activity is its ability to react. How do you use activity series in chemistry? The series is based on empirical data on the ability of a metal to displace hydrogen gas from water and acid. The reactivity series is also known as the activity series of metals. The reactivity series is an ordering of metals from most reactive to least reactive. What is the difference between reactivity and activity series? 
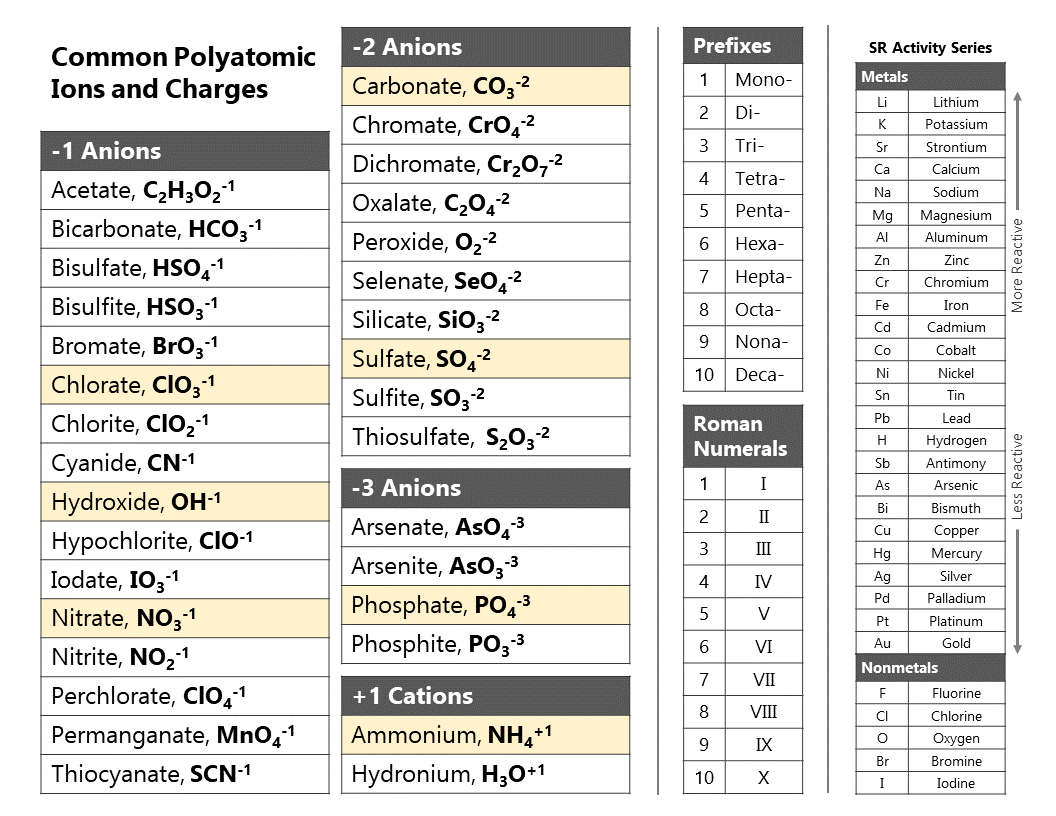
Activity series are generally listed in order of decreasing reactivity. Once organized, an activity series helps us to predict if a reaction will occur when a piece of elemental metal is placed in water, an acid solution, or a solution containing the ion of another metal. What is the importance of activity series? Most reactive metals are on top and least reactive metals are on the botton of the list. The reactivity of metals is determined using single-replacement reaction and the positions are determined based on their reactivity. The activity series of metals is organized based on their reactivity with other metals. How is activity series of metals organized? The elements toward the bottom left corner of the periodic table are the metals that are the most active in the sense of being the most reactive. 
The primary difference between metals is the ease with which they undergo chemical reactions. How do you determine the activity of a metal? The activity series including these elements would be Mg > Zn > H. so zinc is also more active than hydrogen. What is an example of activity series?Īn activity series is a list of substances ranked in order of relative reactivity. It can be used to predict the products in similar reactions involving a different metal. The activity series of metals is an empirical tool used to predict products in displacement reactions and reactivity of metals with water and acids in replacement reactions and ore extraction. What is an activity series and how is it used? The activity series determines the level of reactivity based on how well a certain element can displace hydrogen gas from acidic solutions and water. The activity series is a type of ordering system for elements, which ranks how reactive a certain element is in relation to other elements. In the reaction with a hydrogen-ion source, the metal is oxidized to a metal ion, and the hydrogen ion is reduced to H2. The activity or electromotive series of metals is a listing of the metals in decreasing order of their reactivity with hydrogen-ion sources such as water and acids. 24 What is meant by activity of an ion? What is the activity of a metal? 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
